The Depo-Provera Lawsuit Updates You Need Right Now
The Depo-Provera lawsuit updates most people are searching for comes down to this:
Quick Summary (as of early 2026):
| What’s Happening | Key Details |
|---|---|
| Total lawsuits filed | Over 2,100 in federal court as of February 2026 |
| MDL location | Northern District of Florida, Judge M. Casey Rodgers |
| MDL number | MDL 3140 |
| Core allegation | Pfizer failed to warn U.S. users about brain tumor (meningioma) risk |
| FDA label update | Meningioma warning added in December 2025 |
| Estimated settlements | $275,000 to $1.5 million+ depending on severity |
| Who may qualify | Women who used Depo-Provera for 1+ year and were diagnosed with meningioma |
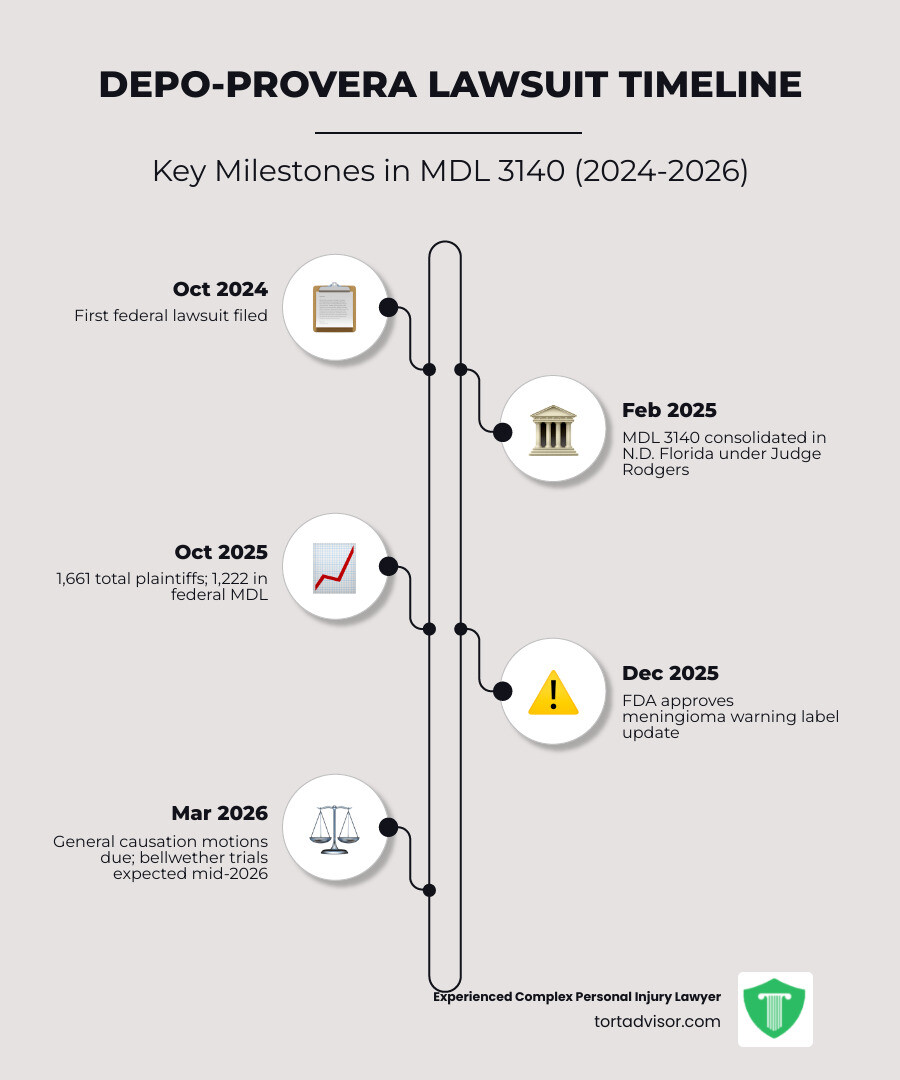
| Next key deadline | General causation motions due March 22, 2026 |
Thousands of women who used the Depo-Provera birth control shot are now suing Pfizer. They say the company knew – or should have known – that the drug raises the risk of developing meningioma brain tumors, but never warned them.
A landmark 2024 study published in the BMJ found that women who used Depo-Provera for more than one year were 5.6 times more likely to develop a meningioma. Despite this, the U.S. warning label remained unchanged for years – even as Canada added a meningioma warning as far back as 2016.
The litigation is moving fast. Case counts grew by 122% in just two months between August and October 2025. Five pilot bellwether trials have been ordered to begin, which could set the stage for a broader settlement.
If you or someone you love used Depo-Provera and later received a brain tumor diagnosis, this guide will walk you through everything happening in the litigation right now.
I’m Mason Arnao, and my background in data systems and internet research has given me a unique lens for tracking complex litigation like the Depo-Provera lawsuit updates – cutting through the noise to surface what actually matters. In this guide, I’ll break down the latest court developments, who qualifies to file, and what potential compensation may look like.
Common Depo-Provera lawsuit updates vocab:
Understanding the Depo-Provera Lawsuit Update and Meningioma Risks
At the heart of every Depo-Provera lawsuit update is a single, terrifying medical term: intracranial meningioma. While these are the most common type of primary brain tumor-accounting for roughly 40% of all central nervous system neoplasms-learning you have a mass growing in your skull is life-altering.
What the Science Says
The link between Depo-Provera (medroxyprogesterone acetate or MPA) and brain tumors isn’t just a legal theory; it is rooted in significant scientific research on progestogen and meningioma risk.
- The 2024 BMJ Study: This massive French study analyzed over 18,000 women who underwent surgery for meningiomas. It revealed that using injectable MPA for more than a year resulted in a 5.6-fold increased risk of developing these tumors.
- The 2025 Cleveland Clinic Study: Published in JAMA Neurology, this study looked at data from over 10 million women. It confirmed a 2.43 relative risk for long-term users, estimating that one additional meningioma occurs for every 1,100 women exposed to the shot.
- The 2025 UBC Study: Researchers at the University of British Columbia found a 3.55-fold increased risk specifically when compared to other forms of birth control.
Why Depo-Provera?
Meningiomas are hormone-sensitive. They often feature high concentrations of progesterone receptors. Because Depo-Provera is a high-dose progestogen-delivered as a 150mg injection every three months-it essentially “feeds” these receptors. Scientists have suspected this link since at least 1983, yet Pfizer is accused of keeping this information off U.S. labels while updating them in other countries.
If you are concerned about your health history, you can read more about the Depo Shot Brain Tumor connection and ask yourself: Does Depo Provera Cause Brain Tumors? The mounting evidence suggests the answer is a resounding yes for many long-term users. For a deeper dive into the data, see this scientific study on MPA and meningioma safety.
Current Status of the Depo-Provera Multidistrict Litigation (MDL 3140)
If you’re looking for a Depo-Provera lawsuit update regarding the “where” and “who,” the answer is the Northern District of Florida. In February 2025, the Judicial Panel on Multidistrict Litigation (JPML) consolidated federal cases into MDL 3140.
The Courtroom Players
The litigation is presided over by Judge M. Casey Rodgers. If that name sounds familiar, it’s because she oversaw the massive 3M earplug litigation, one of the largest mass torts in history. Her experience is vital because this MDL is growing at a breakneck pace.
Discovery and Pfizer’s Defense
We are currently in the heavy “discovery” phase. This is when lawyers from both sides exchange evidence. As of mid-2025, Pfizer had already produced approximately 1 million documents, totaling over 8 million pages.
Pfizer’s primary legal shield has been the “preemption” defense. They argue that because the FDA regulates drug labels, they couldn’t have added a warning without federal permission. However, many legal experts believe this will fail because Pfizer allegedly withheld the very data the FDA would have needed to mandate a warning sooner.
For those tracking the timeline, check out our Depo-Provera Lawsuit Updates 2025 and the Depo-Provera Class Action Lawsuit 2024 summary to see how we got here.
MDL Case Growth Comparison
The surge in filings is one of the most significant aspects of the latest Depo-Provera lawsuit update.
Latest Depo-Provera Lawsuit Updates: Case Counts and Filings
As of February 2026, the number of lawsuits has surpassed 2,100. This reflects a massive influx of women who only recently discovered the link between their past birth control use and their current neurological struggles. These are filed as individual damage claims within the Depo Provera Lawsuits, meaning each woman’s specific medical history and suffering will be evaluated on its own merits.
How to Join the Litigation Following a Depo-Provera Lawsuit Update
Think you might have a claim? You aren’t too late, but you need to act. The discovery rule is your best friend here. Even if you were diagnosed years ago, the “clock” for the statute of limitations typically doesn’t start until you knew (or should have known) that Depo-Provera caused your injury. Since the major studies only came out in 2024 and 2025, most women are still well within their rights to file.
To start, you’ll need to confirm you meet the Depo-Provera Lawsuit Qualifications. If you do, your attorney will help you fill out the Plaintiff Proof of Use/Injury Questionnaire through the BrownGreer MDL Centrality portal. This is a mandatory step to stay active in the MDL. Check your status with our Depo-Provera Lawsuit Eligibility 2025 guide.
Eligibility and Evidence for a Depo-Provera Claim
To be part of the next Depo-Provera lawsuit updates success story, you need more than just a hunch. You need a “paper trail.”
The Minimum Requirements
While every law firm has slightly different intake rules, the general consensus for the strongest cases includes:
- Usage: At least two injections of brand-name Depo-Provera or an authorized generic (like those from Greenstone or Prasco).
- Duration: Most successful claims involve at least one year of use, as the risk multipliers jump significantly after the 12-month mark.
- Diagnosis: A confirmed diagnosis of a cerebral or spinal meningioma.
Essential Evidence
You will need to gather:
- Pharmacy Records: Proving you received the shots.
- Medical Records: Specifically the “op reports” if you had surgery.
- Imaging: MRI or CT scans showing the location and size of the tumor.
- Pathology Reports: To confirm the tumor type (Grade I, II, or III).
For a complete list of what you need, refer to our Depo-Provera Lawsuit Criteria Guide and the Depo-Provera Injury Claim Complete Guide. Understanding the Depo-Provera Severe Side Effects Guide can also help you identify symptoms you might have overlooked in the past.
Potential Settlements and Compensation Tiers
We know what everyone wants to know: “How much is my case worth?” While no global settlement has been reached yet, we can look at similar mass torts and past meningioma malpractice cases to make educated guesses.
The Tiered System
In large MDLs like this, settlements are often divided into “tiers” based on the severity of the injury:
- Tier 1 (Highest Payouts): Women who required multiple brain surgeries, suffered permanent disability (like vision loss or cognitive impairment), or have Grade II or III (atypical or malignant) tumors. These could see over $1 million.
- Tier 2: Women who had a successful single surgery with a Grade I tumor but faced significant recovery time and medical bills. Estimates range from $500,000 to $800,000.
- Tier 3: “Watch and wait” cases where a tumor was found but surgery isn’t yet required, or cases with minor lingering symptoms. These may settle for $100,000 to $300,000.
What Damages Can You Claim?
Compensation isn’t just about the tumor itself; it’s about how your life changed. You can seek money for:
- Medical Expenses: Surgery costs can exceed $700,000.
- Lost Wages: Time missed from work during recovery.
- Pain and Suffering: The emotional toll of a brain tumor diagnosis.
- Loss of Quality of Life: If you can no longer drive, work, or care for your family.
Check out the Depo-Provera Settlement Amounts Lawsuit Guide for more details, or try our Depo Provera Settlement Calculator for a personalized estimate. Our Depo-Provera Lawsuit Compensation Guide 2025 provides even more context on these figures.
Frequently Asked Questions about Depo-Provera Brain Tumors
What are the symptoms of a Depo-Provera brain tumor?
Meningiomas grow slowly, so symptoms often creep up on you. You should watch for:
- Persistent, worsening headaches.
- Changes in vision (blurriness, double vision, or loss of peripheral vision).
- Hearing loss or ringing in the ears.
- Seizures.
- Loss of smell.
- Weakness in arms or legs.
If you have a history of Depo-Provera use and experience these, consult a neurologist immediately. You can read a full list of Depo Provera Brain Tumor Symptoms here. These are considered Depo-Provera severe side effects.
Has the FDA issued a recall for Depo-Provera?
No, as of early 2026, Depo-Provera has not been recalled. However, the litigation has forced a major change. In December 2025, the FDA finally approved a new warning label that includes the risk of meningiomas. This follows years of “black box” warnings for bone density loss, which you can read about in our Depo-Provera Lawsuit Side Effects Guide. This label change is a huge win for the Birth Control Shot Lawsuit movement, as it essentially acknowledges the risk exists.
What is the statute of limitations for filing a claim?
This is the most common question in any Depo-Provera lawsuit update. The deadline varies by state (usually 2 to 3 years), but the discovery rule generally protects you. Because the scientific link wasn’t widely publicized until 2024, many courts will rule that your “timer” didn’t start until then.
However, if Pfizer is found guilty of fraudulent concealment (hiding the risk), those deadlines could be extended even further through “equitable tolling.” Don’t guess on this-consult our Depo-Provera Lawsuit Payout Date Guide and the Depo-Provera Lawsuit News Ultimate Guide for help.
Conclusion
The Depo-Provera lawsuit update landscape is shifting every single week. With over 2,100 cases now pending and the first bellwether trials on the horizon for 2026, the window for holding Pfizer accountable is wide open.
At Tort Advisor, we believe no woman should have to choose between effective birth control and her long-term neurological health. We connect clients with top-rated specialty attorneys who have proven results in complex pharmaceutical litigation. Whether you are in Alabama, California, Florida, or any of our 50+ service locations, we are here to ensure you aren’t fighting this battle alone.
If you’re ready to take the next step, read our Depo Provera Lawsuit Complete Guide or contact us for a free case review today. Your health and your rights are worth the fight.
Free Confidential Case Evaluation
Complete the short form below to get an immediate FREE case review with an expert in your specific claim. Don't wait, your case could be time sensitive to file a claim.
Related Posts
Breaking Depo-Provera lawsuit update: Pfizer files Daubert motion in the meningioma MDL ahead of June 24–26 causation hearings. 3,790 plaintiffs, settlement estimates $100K–$1.5M, who qualifies in 2026. Call 1 (855) 664-8713 for a free case review.
Discover what to do after a Maryland car accident: immediate steps, insurance laws, claims, and legal help for commuters on I-95.
Discover the average payout for cancer misdiagnosis: $450K median settlements. Learn factors, real cases & how to claim compensation today.
Learn about the recall on gummy bear implants, BIA-ALCL risks, affected models, symptoms, lawsuits & compensation. Get expert legal guidance now!
Hire a defective product injury lawyer to fight for compensation after a defect causes harm. Learn strict liability, defects & claims.
Discover PFAS lawsuit settlement amounts per person kidney cancer: $150K-$500K estimates, eligibility, timelines & claim guide. Get started now!