

A meningioma brain tumor is one of the most serious and life-altering medical conditions linked to long-term use of Depo-Provera (medroxyprogesterone acetate), Pfizer’s injectable birth control contraceptive. If you or someone you love used Depo-Provera and was later diagnosed with a meningioma, you may be entitled to significant financial compensation through the Depo-Provera meningioma lawsuit currently active in MDL 3140. As of May 2026, more than 3,769 victims have already filed claims — and the filing window may be closing. Call 1 (855) 664-8713 or start your free Depo-Provera case evaluation online today.
My name is Mason Arnao, and my background in data systems, technology infrastructure, and digital advocacy has given me a deep understanding of how the Depo-Provera meningioma lawsuit 2026 affects real people navigating complex legal and medical systems. As publisher of TortAdvisor.com and President of Waypoint Software — a leading digital firm with over 20 years of experience — I’ve built systems that connect people with critical information. In this guide, I’ll break down everything you need to know about meningioma brain tumors, Depo-Provera’s alarming connection to them, and how to take legal action.
📞 Free Confidential Case Evaluation — No Fee Unless You Win
Call Us to Speak With a Depo-Provera Meningioma Expert Now
Open 24/7 · Completely Confidential · No Upfront Cost
📋 Table of Contents
- What Is a Meningioma Brain Tumor?
- Depo-Provera and Meningioma: The Alarming Scientific Link
- Symptoms, Diagnosis, and Grading of Meningioma
- Depo-Provera Lawsuit 2026: MDL 3140 Update
- Who Qualifies for a Depo-Provera Lawsuit?
- Depo-Provera Settlement Amounts 2026
- Related Mass Tort Lawsuits
- How to File a Depo-Provera Meningioma Lawsuit
- Frequently Asked Questions
- About the Author
What Is a Meningioma Brain Tumor?
A meningioma brain tumor is a tumor that grows from the meninges — the three layers of protective membrane that encase the brain and spinal cord. Unlike glioblastomas or other aggressive cancers, most meningiomas are classified as Grade I (benign), meaning they grow slowly and rarely spread. However, “benign” is deeply misleading when it comes to meningiomas: because they form inside the skull, even a slow-growing tumor can compress critical brain structures, causing permanent neurological damage, disability, and in severe cases, death.
Furthermore, approximately 10–15% of meningiomas are Grade II (atypical) and 1–3% are Grade III (malignant), both of which carry significantly more aggressive growth patterns and poorer prognoses. The American Cancer Society reports roughly 34,000 new meningioma cases diagnosed annually in the United States. Women are diagnosed at nearly twice the rate of men — a disparity that researchers have increasingly linked to hormonal exposure, specifically progestin-containing contraceptives like Depo-Provera.
Meningiomas can form anywhere along the meninges — above the brain, at the base of the skull, along the spinal cord, or near the optic nerves. The location determines the severity of symptoms and the difficulty of surgical removal. For example, a meningioma near the optic nerve can cause progressive vision loss or blindness, while one near the motor cortex can cause weakness, paralysis, or seizures. The Mayo Clinic notes that treatment options — including surgery, radiation, and stereotactic radiosurgery — carry their own significant risks, especially when tumors are located in surgically sensitive areas.
What makes Depo-Provera-related meningiomas particularly alarming is that they are preventable. Growing scientific evidence — and now mounting legal action — shows that Pfizer knew or should have known about meningioma risk long before patients were warned. The result is thousands of women who developed life-altering brain tumors from a contraceptive that was never adequately labeled with this critical safety warning. The Depo-Provera meningioma lawsuit 2026 represents the legal reckoning for this corporate negligence.
Understanding what a meningioma is, how it develops, and how it connects to Depo-Provera is the first critical step in building your case. Specifically, if you used Depo-Provera (medroxyprogesterone acetate) for 12 months or more and were subsequently diagnosed with a meningioma, you may have a viable personal injury lawsuit. To understand your rights and estimate what your claim may be worth, use our free Depo-Provera settlement calculator or call 1 (855) 664-8713 now.
Depo-Provera and Meningioma: The Alarming Scientific Link
The connection between Depo-Provera and meningioma brain tumors is no longer a theory — it is a scientifically validated, FDA-acknowledged reality. Depo-Provera contains medroxyprogesterone acetate (MPA), a synthetic progestin that mimics progesterone but is significantly more potent. Research has established that meningioma tumors express progesterone receptors, meaning hormonal signals — including synthetic progestins like MPA — can directly stimulate meningioma growth.
The landmark study that triggered the current wave of Depo-Provera litigation was a 2024 French epidemiological study published in the British Medical Journal (BMJ) analyzing over 18,000 meningioma cases. The study found that women who used high-potency progestins — including medroxyprogesterone acetate (the active ingredient in Depo-Provera) — had a dramatically elevated risk of developing intracranial meningiomas compared to non-users. Notably, the risk increased proportionally with duration of use: women who used Depo-Provera for 3 or more years faced the highest relative risk elevations.
The FDA responded to this emerging body of evidence by requiring Pfizer to update Depo-Provera’s drug label with a meningioma warning. The updated label acknowledged that prolonged use of medroxyprogesterone acetate is associated with an increased risk of meningioma and recommended that healthcare providers consider discontinuing Depo-Provera in patients diagnosed with meningioma. This FDA label update is a critical piece of evidence in the ongoing Depo-Provera meningioma lawsuit 2026 — it confirms that the risk was real, scientifically established, and warrants a warning that was absent for years during which millions of women were prescribed the drug.
The biological mechanism is well understood: meningiomas contain progesterone receptors in approximately 60–80% of cases, according to research published by the National Institutes of Health (NIH). MPA, being a potent synthetic progestin, binds to these receptors and stimulates cellular proliferation. The longer a woman is exposed to exogenous MPA — as with quarterly Depo-Provera injections over years or decades — the greater the cumulative hormonal stimulation of any existing progesterone receptor-positive meningioma cells.
Importantly, prior research had already flagged similar risks with other high-potency progestins used in Europe, particularly cyproterone acetate and chlormadinone acetate, both of which have been the subject of litigation and regulatory action in France and elsewhere. The Depo-Provera litigation argues that Pfizer had constructive knowledge of meningioma risk through this related body of progestin research and failed to act on it — a core element of product liability claims in MDL 3140.
Additionally, clinical evidence shows that meningiomas shrink after discontinuation of progestin use in many cases — further confirming the hormonal mechanism. This regression-upon-cessation phenomenon is powerful evidence both scientifically and legally, directly linking the drug to tumor growth. If you used Depo-Provera and have been diagnosed with a meningioma, the science strongly supports your claim. Visit our Depo-Provera lawsuit guide to learn more about how to proceed.

📞 Free Confidential Case Evaluation — No Fee Unless You Win
Call Us to Speak With a Depo-Provera Meningioma Expert Now
Open 24/7 · Completely Confidential · No Upfront Cost
Symptoms, Diagnosis, and Grading of Meningioma
One of the most challenging aspects of meningioma is that symptoms often develop slowly and can be easily dismissed or misattributed to other conditions for years. However, as the tumor grows and compresses surrounding brain tissue, symptoms become increasingly severe and disabling. Understanding the symptoms, diagnosis process, and grading of meningioma is essential both for getting appropriate medical treatment and for documenting your Depo-Provera meningioma lawsuit claim.
Common meningioma symptoms include headaches (often worst in the morning), seizures, vision changes or double vision, hearing loss or ringing in the ears, memory problems and cognitive decline, weakness or numbness in the arms or legs, speech difficulties, and personality or behavioral changes. Notably, many of these symptoms are progressive — they worsen gradually over months or years, which is consistent with the slow growth of hormone-stimulated meningiomas. Many women report that their symptoms were dismissed for years before a meningioma was finally identified on imaging.
Meningioma diagnosis typically begins with an MRI (magnetic resonance imaging) with contrast, which is the gold standard for identifying and characterizing these tumors. A CT scan may also be used, particularly in emergency settings. In cases where surgery or biopsy is performed, pathological tissue examination confirms the grade. The Mayo Clinic notes that many low-grade meningiomas are found incidentally during imaging for unrelated symptoms — however, discovery does not diminish the harm or the legal claim.
The WHO grading system for meningiomas classifies tumors as follows: Grade I (benign) — approximately 80% of cases, slow growth, good prognosis with complete surgical removal; Grade II (atypical) — approximately 15–18% of cases, higher recurrence rate, more aggressive treatment required; Grade III (malignant/anaplastic) — approximately 1–3% of cases, aggressive growth, poor prognosis, requiring aggressive treatment including surgery, radiation, and possible chemotherapy. Even Grade I meningiomas can cause permanent disability depending on location, and the treatments themselves — brain surgery and radiation — carry serious risks of complications including cognitive deficits, infection, bleeding, and stroke.
From a legal perspective, meningioma diagnosis, grade, treatment history, and documented impact on quality of life are all critical components of your Depo-Provera meningioma lawsuit 2026 claim. Higher-grade tumors, surgeries, and ongoing disability generally result in higher compensation awards. Our Depo-Provera settlement calculator uses your specific diagnosis details, treatment history, and severity of impact to provide a personalized estimate of your claim’s potential value. Consequently, gathering complete medical records — including your Depo-Provera prescription history and all imaging and surgical reports — is one of the most important steps you can take right now.
Depo-Provera Lawsuit 2026: MDL 3140 Update
The Depo-Provera meningioma lawsuit 2026 is consolidated in MDL 3140, formally titled In re: Depo-Provera (Medroxyprogesterone Acetate) Products Liability Litigation, presided over in the Northern District of Illinois. As of May 2026, the MDL includes more than 3,769 active cases, making it one of the most significant pharmaceutical mass tort actions currently active in the federal court system. The defendant — Pfizer Inc. and its subsidiary Pharmacia — faces allegations that it failed to adequately warn patients and physicians about the meningioma risk associated with long-term Depo-Provera use.
The MDL consolidation is significant for plaintiffs for several reasons. First, it allows individual cases to proceed through coordinated pretrial discovery, which means that evidence gathered regarding Pfizer’s internal knowledge of meningioma risk — including internal communications, clinical trial data, and regulatory submissions — benefits all plaintiffs collectively. Second, MDL plaintiffs typically retain their right to individual trials rather than being bound to a class-wide settlement, which can result in substantially higher individual compensation for severe cases.
The litigation has reached several critical milestones in 2026. The court is currently overseeing Daubert hearings — proceedings in which the admissibility of expert scientific testimony is evaluated by the judge. The outcome of these hearings will significantly shape the trajectory of the litigation: if the court accepts plaintiffs’ expert testimony linking Depo-Provera to meningioma, the path to bellwether trials and eventual global settlement will be significantly advanced. The Judicial Panel on Multidistrict Litigation website provides official docket updates for MDL 3140.
Legal analysts estimate a potential global settlement fund in the range of $1.5 billion to $3 billion or more, based on comparable pharmaceutical MDL resolutions and the severity of injuries involved. However, individual settlement amounts will vary significantly based on each plaintiff’s specific circumstances — duration of Depo-Provera use, tumor grade, treatments undergone, and documented impact on daily life and earning capacity. Notably, some of the strongest cases — Grade III malignant meningiomas requiring multiple surgeries — may yield individual awards well above the average settlement tier. For a personalized estimate, visit our free Depo-Provera settlement calculator.
Furthermore, the statute of limitations in most states is 2–3 years from the date of meningioma diagnosis or from the date you reasonably discovered the connection between Depo-Provera and your tumor. This deadline is not flexible — once it passes, your right to compensation is permanently forfeited. If you were diagnosed with meningioma and used Depo-Provera, it is critically important that you contact an attorney immediately. Visit our complete Depo-Provera lawsuit guide or call 1 (855) 664-8713 to speak with a case specialist now.
Who Qualifies for a Depo-Provera Meningioma Lawsuit?
Not every Depo-Provera user and not every meningioma patient qualifies for the current litigation — however, the eligibility criteria are straightforward and can be assessed in a free consultation. Based on the current MDL 3140 bellwether criteria and plaintiff intake standards used by leading mass tort firms, the following are the primary eligibility requirements for the Depo-Provera meningioma lawsuit 2026:
- Depo-Provera use: You received injections of Depo-Provera (medroxyprogesterone acetate) for at least 12 consecutive months at any point in your lifetime
- Meningioma diagnosis: You were diagnosed with a meningioma brain tumor (any grade — Grade I, II, or III) confirmed by MRI, CT, or pathological examination
- Causal timeline: Your Depo-Provera use preceded your meningioma diagnosis — the drug use came before the tumor was found
- Within statute of limitations: Your meningioma diagnosis was within the applicable statute of limitations for your state (typically 2–3 years from diagnosis or discovery of the Depo-Provera link)
- U.S. resident: You are a resident of any U.S. state — there is no geographic restriction within the United States
- No prior settlement: You have not previously settled a Depo-Provera meningioma claim
Cases with Grade II or Grade III meningiomas, surgical resection, radiation treatment, or significant documented disability typically qualify for the highest compensation tiers. However, even Grade I meningiomas that are being monitored (watchful waiting) may still qualify, particularly if the tumor has caused documented symptoms or has required treatment. Additionally, cases involving deceased family members who used Depo-Provera and developed meningioma may qualify as wrongful death claims, potentially with even higher compensation for surviving family members.
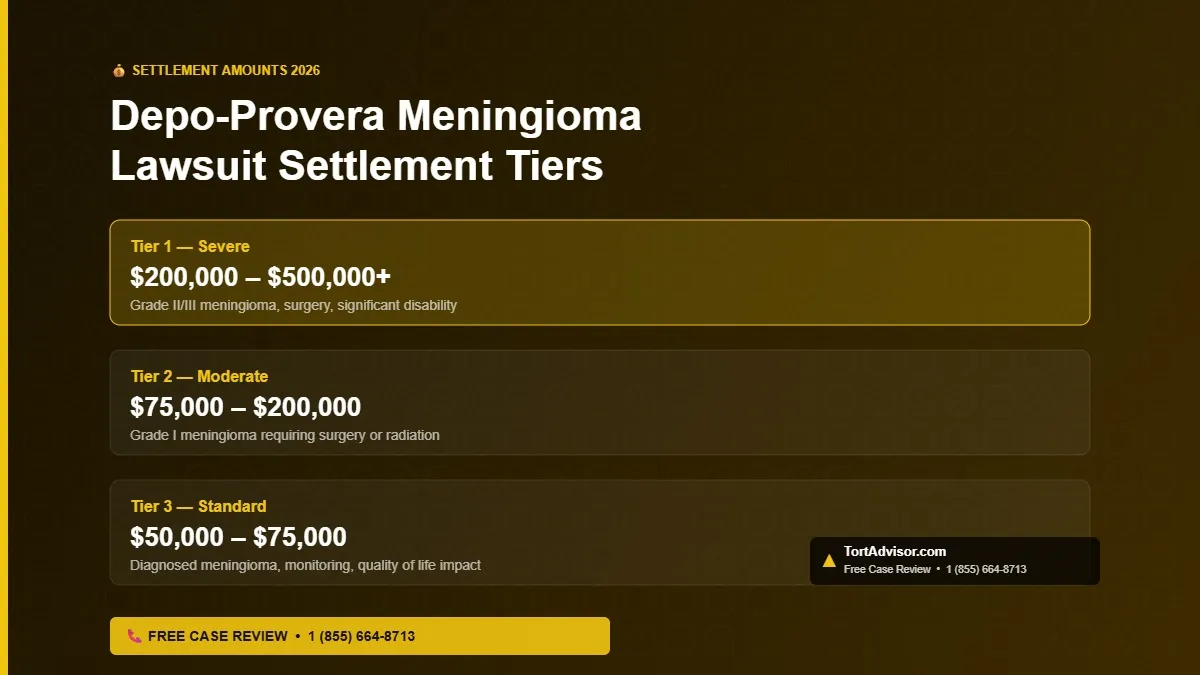
Depo-Provera Meningioma Lawsuit Settlement Amounts 2026
One of the most common questions we receive is: “How much is a Depo-Provera meningioma lawsuit worth?” The honest answer is that settlement amounts will vary significantly based on each individual’s specific medical history, the severity of their meningioma, the treatments they underwent, and the documented impact on their life and earning capacity. That said, based on comparable pharmaceutical MDL settlements, expert projections, and the specific injury profile of Depo-Provera meningioma victims, the following tiers provide a reasonable framework for understanding potential compensation ranges.
It is important to note that these are estimated ranges only — not guarantees. No attorney can promise a specific outcome, and final settlement amounts will depend on the global MDL resolution, individual case strength, and Pfizer’s litigation strategy. Nevertheless, the following tiers reflect widely cited industry estimates and are consistent with outcomes in comparable pharmaceutical mass torts:
| Settlement Tier | Estimated Range | Qualifying Factors |
|---|---|---|
| Tier 1 — Severe/Catastrophic | $200,000 – $500,000+ | Grade II/III meningioma, multiple surgeries, radiation, permanent disability, significant lost wages |
| Tier 2 — Moderate | $75,000 – $200,000 | Grade I meningioma with surgery or radiation, documented symptoms, quality of life impact |
| Tier 3 — Standard | $50,000 – $75,000 | Grade I meningioma under watchful waiting, confirmed diagnosis, some documented symptoms |
| Wrongful Death | $300,000 – $1,000,000+ | Death caused or contributed to by Depo-Provera meningioma; filed by surviving family members |
To get a personalized estimate based on your specific situation, use our free Depo-Provera settlement calculator. It takes under two minutes and provides an immediate estimate. Additionally, we encourage you to review our mass tort settlement amounts page for comparable case values across other pharmaceutical and product liability MDLs.
Related Mass Tort Lawsuits You May Qualify For
If you or a family member have been affected by other dangerous products, defective drugs, or corporate negligence, you may have additional legal options worth exploring. Our attorneys handle a wide range of personal injury lawsuits nationwide, including:
- Hair relaxer cancer lawsuit — chemical hair straighteners linked to uterine cancer and endometrial cancer
- Ozempic gastroparesis lawsuit — GLP-1 diabetes drugs linked to severe stomach paralysis
- Talcum powder lawsuit — Johnson’s Baby Powder and ovarian cancer, mesothelioma
- PFAS cancer lawsuit — forever chemicals in drinking water linked to kidney, bladder, and thyroid cancer
- Breast implant lawsuit — textured implants linked to BIA-ALCL lymphoma
- Free settlement calculators for all case types
Each case is handled on a no-fee-unless-you-win basis. Use our free settlement calculators to estimate what your claim may be worth, or call 1 (855) 664-8713 for a free confidential evaluation.
How to File a Depo-Provera Meningioma Lawsuit in 2026
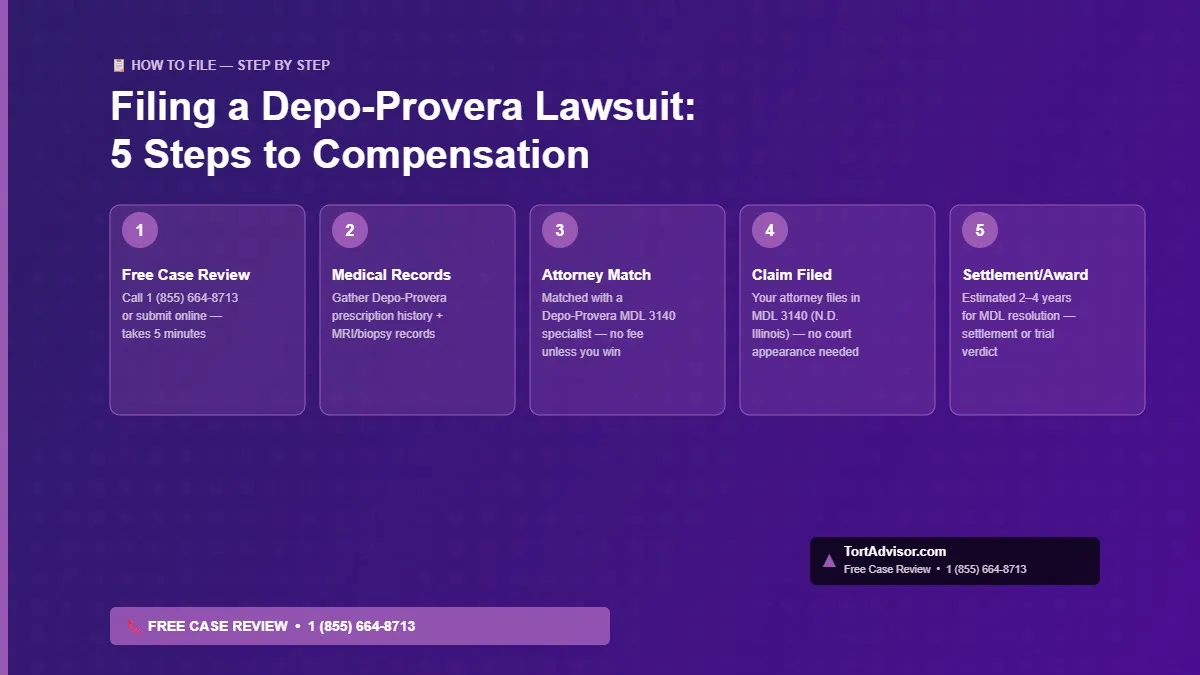
Filing a Depo-Provera meningioma lawsuit is far simpler than most victims realize — and you do not need to navigate the legal system alone. The entire process can be completed from home, with no upfront costs and no out-of-pocket fees of any kind. Here is a step-by-step overview of how the Depo-Provera claims process works in 2026:
- Free Case Evaluation (Day 1): Call 1 (855) 664-8713 or start your case evaluation online. A case specialist will ask a few basic questions about your Depo-Provera use and meningioma diagnosis to confirm preliminary eligibility. This call takes approximately 5–10 minutes and is completely confidential.
- Medical Records Gathering (Week 1–2): Your attorney’s team will assist you in requesting and organizing your medical records, including Depo-Provera prescription and injection records, MRI or CT imaging reports, surgical and pathology reports, radiation treatment records, and any documentation of disability or reduced quality of life.
- Attorney Match and Retainer (Week 2–3): You will be matched with a mass tort attorney specializing in MDL 3140 Depo-Provera litigation. You sign a contingency fee agreement — meaning the attorney only gets paid if and when you receive a settlement or verdict. There are zero upfront costs.
- Claim Filed in MDL 3140 (Month 1–2): Your attorney files your individual complaint in the Northern District of Illinois MDL. You do not need to appear in court at this stage. All filings and communications are handled by your legal team.
- Discovery and Bellwether Process (Month 3–24): Your case participates in MDL-wide discovery and, if selected, may serve as a bellwether trial to establish case value precedents. Most individual plaintiffs are not selected for bellwether trials.
- Settlement or Verdict (Estimated 2–4 Years): Based on MDL precedent, a global settlement is expected within 2–4 years of filing, though some cases may proceed to trial for higher individual awards. Once a settlement is reached, your attorney will present the offer and advise whether to accept it.
Estimate Your Depo-Provera Settlement Amount
Before you call or submit your information, you may want to get an initial estimate of what your Depo-Provera meningioma lawsuit claim could be worth. Our free calculator uses your diagnosis grade, treatment history, and duration of Depo-Provera use to generate a personalized compensation estimate. Use our free Depo-Provera settlement calculator — it takes under two minutes.
Frequently Asked Questions: Depo-Provera Meningioma Lawsuit 2026
What is a meningioma brain tumor and how is it linked to Depo-Provera?
A meningioma is a tumor that grows from the meninges, the protective membranes surrounding the brain and spinal cord. Scientific studies, including a landmark 2024 BMJ study, found that medroxyprogesterone acetate — the active ingredient in Depo-Provera — significantly increases meningioma risk because meningioma cells contain progesterone receptors that are stimulated by synthetic progestins. The longer a woman uses Depo-Provera, the greater the accumulated risk. The FDA has acknowledged this link and required Pfizer to update Depo-Provera’s drug label with a meningioma warning.
Is it too late to file a Depo-Provera meningioma lawsuit in 2026?
Most states allow 2–3 years from the date of your meningioma diagnosis — or from the date you discovered the Depo-Provera connection — to file a lawsuit. However, statutes of limitations vary by state and can be impacted by when you first learned of the drug’s role in your tumor. Acting quickly is critical: once the deadline passes, your right to file is permanently lost. Call 1 (855) 664-8713 immediately for a free evaluation to determine your specific deadline. Many victims still have time to file as of May 2026, but the window is closing for early diagnoses.
How much is a Depo-Provera meningioma lawsuit worth in 2026?
Estimated Depo-Provera meningioma lawsuit settlement amounts range from approximately $50,000 for Grade I cases under watchful waiting, to $75,000–$200,000 for cases involving surgery or radiation, to $200,000–$500,000 or more for severe Grade II/III meningiomas with significant disability. Wrongful death cases may yield $300,000–$1,000,000+. These are estimates based on comparable MDL settlements — individual results vary. Use our free Depo-Provera settlement calculator for a personalized estimate.
Who qualifies for a Depo-Provera meningioma lawsuit?
To qualify for the Depo-Provera meningioma lawsuit, you generally must have: (1) used Depo-Provera (medroxyprogesterone acetate) for at least 12 consecutive months; (2) received a confirmed meningioma diagnosis (any grade) via MRI, CT, or pathology; (3) used Depo-Provera before your diagnosis; and (4) be within your state’s statute of limitations. U.S. residents of any state may qualify. Family members of deceased Depo-Provera users may qualify for wrongful death claims.
How long does a Depo-Provera meningioma lawsuit take to resolve?
MDL 3140 is currently in the active pretrial phase as of May 2026. Based on comparable pharmaceutical MDL timelines — including the Roundup MDL and Talcum Powder MDL — a global settlement or bellwether trial verdict is generally expected within 2–4 years of MDL formation. MDL 3140 was established in 2024, suggesting a potential resolution in the 2026–2028 timeframe, though complex litigation timelines are inherently uncertain. Filing sooner rather than later positions your claim for the earliest possible resolution.
What happened with the Depo-Provera Daubert hearings in 2026?
As of May 2026, MDL 3140 is actively proceeding through Daubert hearings — the process by which the judge evaluates whether plaintiffs’ expert scientific testimony linking Depo-Provera to meningioma meets the standard for admissibility at trial. The Daubert phase is a critical gateway: favorable rulings will strengthen all plaintiffs’ positions and accelerate the path toward bellwether trials and global settlement negotiations. The case docket is available through the federal court system and updated regularly at the JPML website. Our Depo-Provera lawsuit guide is updated regularly with case news.
Do I need a lawyer or can I file a Depo-Provera lawsuit on my own?
While it is theoretically possible to file pro se (without an attorney), it is strongly inadvisable in MDL litigation, which involves complex procedural rules, expert witness requirements, and case management orders. Mass tort attorneys handle all aspects of your case on a contingency fee basis — you pay nothing unless you win. Working with a qualified Depo-Provera meningioma attorney gives your case the best chance of maximum compensation and ensures you meet all filing deadlines. Call 1 (855) 664-8713 for a free, no-obligation consultation today.
⚠️ Time-Sensitive — Statutes of Limitations Apply
Call Us to Speak With a Depo-Provera Meningioma Expert Now
1 (855) 664-8713
Start Your Free Case Evaluation Online →
Free Confidential Consultation · Open 24/7 · No Fee Unless You Win
About the Author
My name is Mason Arnao, and my background in data systems, technology infrastructure, and digital advocacy has given me a deep understanding of how the Depo-Provera meningioma lawsuit 2026 affects real people navigating complex legal and medical systems. As publisher of TortAdvisor.com and President of Waypoint Software — a leading digital firm with over 20 years of experience — I’ve built systems that connect people with critical information. In this guide, I’ve broken down everything you need to know to understand your rights and take action against Pfizer for the harm caused by Depo-Provera.
Free Confidential Case Evaluation
Complete the short form below to get an immediate FREE case review with an expert in your specific claim. Don't wait, your case could be time sensitive to file a claim.
Related Posts
Institutional sexual abuse lawsuit 2026: Churches, schools, hospitals, detention centers. Who qualifies, settlement amounts, how to file. Free confidential review. 1 (855) 664-8713.
SSDI claims 2026: Complete guide to winning Social Security Disability benefits. Who qualifies, how to appeal denials, free case review. 1 (855) 664-8713.
Who qualifies for WTC VCF claims 2026? FDNY, NYPD, first responders — 50+ cancers covered. Tax-free awards. Free review. 1 (855) 664-8713.
World Trade Center lawsuit 2026: 9/11 VCF guide for FDNY, NYPD, first responders. 50+ cancers covered. Free VCF review. 1 (855) 664-8713.
Lyft sexual assault lawsuit 2026: 2,000+ reported incidents. Who qualifies, settlement amounts, how to sue Lyft. Free confidential review. 1 (855) 664-8713.
Uber sexual assault lawsuit 2026: $8.5M verdict won. Who qualifies, settlement amounts, how to file. Free confidential case review. 1 (855) 664-8713.