What the Recall on Gummy Bear Implants Means for You — and Your Legal Rights
The recall on gummy bear implants is one of the most significant medical device safety actions in recent history, and if you have Allergan BIOCELL textured implants, you may have legal rights you don’t even know about yet.
Here’s what you need to know right now:
- What was recalled: In July 2019, Allergan recalled all BIOCELL textured breast implants worldwide — including the Natrelle 410 “gummy bear” implants — at the FDA’s request.
- Why: These implants were linked to a rare cancer called Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL). Allergan implants were found to carry six times the cancer risk of other textured implants.
- How many cases: As of 2023, the FDA had recorded 1,264 BIA-ALCL cases worldwide — 85% linked to Allergan implants — and 63 deaths.
- Do you need surgery now? The FDA does not recommend removing implants if you have no symptoms. But you should monitor for warning signs like swelling, lumps, or breast pain.
- Can you get compensation? Yes. Lawsuits have been filed against Allergan, and affected patients may be eligible for compensation — even if they haven’t been diagnosed with BIA-ALCL.
This is not just a medical issue. For thousands of women, it has also become a financial and legal nightmare. Allergan offered to cover the cost of replacement implants — but not the cost of removal surgery, unless a BIA-ALCL diagnosis was confirmed. Many women were left paying tens of thousands of dollars out of pocket.
One cancer survivor described her eight-year struggle to have her recalled implants removed, blocked at every turn by insurers and surgeons citing FDA guidelines that didn’t recommend routine removal. Her story is not unique.
My name is Mason Arnao, and my background in data systems, research, and digital advocacy has given me a deep understanding of how product recalls — including the recall on gummy bear implants — affect real people navigating complex legal and medical systems. In this guide, I’ll break down everything you need to know to understand your rights and take action.
Understanding the Recall on Gummy Bear Implants and BIA-ALCL
When we talk about the recall on gummy bear implants, we are specifically looking at a massive safety intervention triggered by the FDA in 2019. This wasn’t just a minor paperwork error; it was a response to a direct link between a specific manufacturing process and a rare form of cancer.
The FDA requested the Allergan recall after data showed that Allergan BIOCELL textured implants were disproportionately represented in cases of Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL). To put it bluntly, while BIA-ALCL is a rare T-cell lymphoma of the immune system, it isn’t “random.” It has a clear preference for textured surfaces.
What Defines a Gummy Bear Implant?
The term “gummy bear” is a bit of marketing genius used by surgeons and manufacturers, but medically, these are known as “form-stable” or “highly cohesive” silicone gel implants. They earned their nickname because the silicone inside is so thick and stable that if you were to cut the implant in half, the gel would stay put—much like the candy.
These implants are designed to maintain a specific shape (often a teardrop) and are almost always textured to prevent them from rotating inside the breast. It is this specific combination—the highly cohesive gel and the rough BIOCELL texture—that sits at the heart of the gummy bear implants recall. While they were praised for their natural look, the “velcro-like” surface intended to keep them in place turned out to be a biological trigger for some patients.
The Link Between BIOCELL Texture and Cancer
So, why did these specific implants become the focus of a global recall? The answer lies in the “lost salt” technique Allergan used to create the BIOCELL texture. This process creates deep, irregular pores on the implant shell.
There are two primary theories on why this causes cancer:
- The Bacterial Biofilm Theory: Those deep pores can trap bacteria, creating a “biofilm” that leads to chronic, low-grade inflammation. Over years, this persistent immune response can trigger the mutation of T-cells into lymphoma.
- The Silicone Particle Theory: The mechanical friction of the rough texture against the body’s tissue may cause microscopic silicone particles to rub off, leading to chronic inflammation.
Whatever the exact mechanism, the statistics are hard to ignore. If you are concerned about your diagnosis, speaking with a breast implant cancer lawyer can help you understand if your specific manufacturer is liable.
| Feature | Smooth Implants | Textured (BIOCELL) Implants |
|---|---|---|
| BIA-ALCL Risk | Negligible/None identified | ~6x higher than other brands |
| Surface | Smooth, slippery | Rough, “velcro-like” |
| Common Use | Cosmetic augmentation | Reconstruction/Form-stable |
| Recall Status | Generally safe | Voluntarily recalled (2019) |
Identifying Recalled Models and Symptoms
If you had surgery between 2006 and 2019, there is a significant chance you have one of the models included in the recall on gummy bear implants. It is vital to check your “implant card” or medical records to see if your devices match the list below.
Specific Models Affected by the Recall on Gummy Bear Implants
The recall wasn’t limited to just one “type” of implant; it covered a wide range of Allergan’s BIOCELL product line. This includes:
- Natrelle 410 Highly Cohesive Anatomically Shaped (The classic “Gummy Bear”)
- Natrelle Silicone-Filled Textured Implants (Styles 110, 115, 120, TRL, TRLP, etc.)
- Natrelle Saline-Filled Textured Implants (Styles 163, 168, 363, 468)
- Natrelle Inspira (Textured models)
- Tissue Expanders: Used in the first stage of reconstruction, specifically the Natrelle 133 Plus and Natrelle 133 with Suture Tabs.
Even as we move through breast implant recall 2023 updates, these specific models remain the primary concern for legal and medical monitoring.
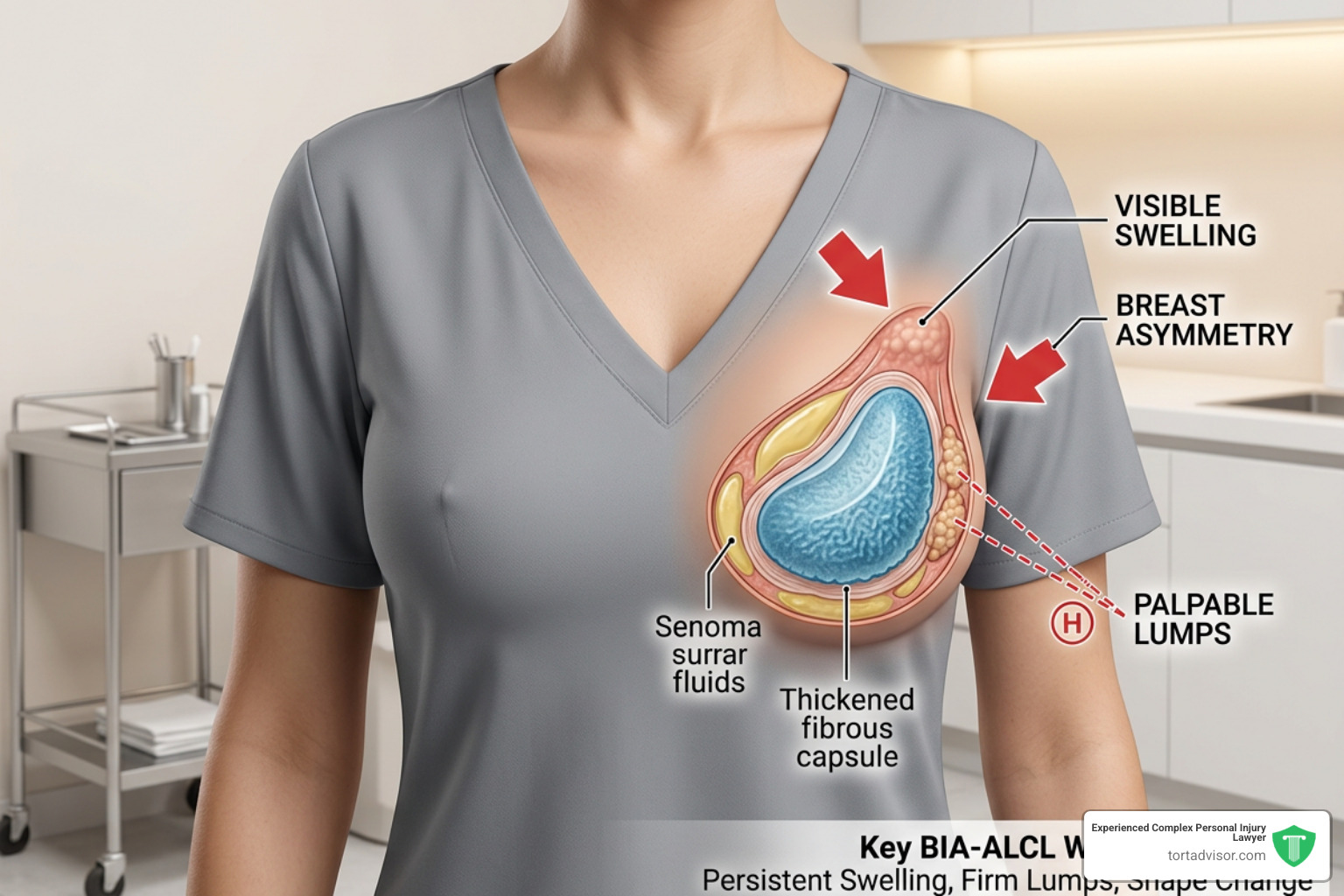
Recognizing Symptoms of BIA-ALCL and Complications
BIA-ALCL is not breast cancer; it is a cancer of the immune system found in the fluid (seroma) or scar tissue (capsule) surrounding the implant. Symptoms usually appear 3 to 14 years after the initial surgery. We advise all patients to watch for:
- Delayed Seroma: Sudden swelling or fluid collection in one breast long after the surgical site has healed.
- Lumps or Masses: Feeling a hard lump in the breast or armpit.
- Capsular Contracture: A noticeable hardening or tightening of the breast.
- Asymmetry: One breast suddenly looking much larger or a different shape than the other.
- Systemic Symptoms: Unexplained fatigue, skin rashes, or weight loss.
If you experience these, it is crucial to seek medical attention immediately. You may also want to browse breast-implant-lawsuits to see how other women with these symptoms have navigated their claims.
Legal Rights and Compensation for Affected Patients
When a medical device manufacturer puts a product on the market that carries a six-fold higher risk of cancer than its competitors, they may be held liable under product liability laws. For those affected by the recall on gummy bear implants, the legal path is about more than just a refund—it’s about accountability for medical bills, future monitoring, and emotional distress.
Filing a Lawsuit After the Recall on Gummy Bear Implants
Many women find themselves in a “catch-22.” They have a recalled, potentially carcinogenic product in their bodies, but their insurance won’t pay for removal because they aren’t “sick yet.” Meanwhile, Allergan’s initial recall offer only covered the cost of new implants, leaving patients to foot the bill for surgeon fees, anesthesia, and operating room costs—which can exceed $10,000.
By filing an Allergan litigation claim, we can seek to recover:
- Surgical Costs: The full price of explantation (removal) and capsulectomy.
- Medical Monitoring: The cost of regular MRIs and physician visits to check for cancer.
- Pain and Suffering: Compensation for the anxiety of living with a “ticking time bomb.”
- Lost Wages: Time taken off for surgeries and recovery.
For a better idea of what your claim might be worth, you can use a breast-implant-settlement-calculator or consult a breast-implant-class-action-lawsuit-guide.
Compensation for Silicone Migration and Toxicity
Beyond the cancer risk, “gummy bear” implants are also under scrutiny for “gel bleed” or silicone migration. A study of 389 women found that nearly 99% had silicone particles in their tissues, and over 86% showed migration outside the breast capsule.
This “bleeding” of silicone can cause a systemic inflammatory response, often referred to by patients as “Breast Implant Illness” (BII). Symptoms can include joint pain, brain fog, and lymph node involvement. If you are suffering from these issues, a breast implant lawsuit silicone toxicity claim may be your best route to securing the funds needed for a “total capsulectomy” (removing the implant and the entire scar tissue bag).
Medical Recommendations and Safer Alternatives
The FDA’s current guidance on breast implants is clear: if you don’t have symptoms, they do not recommend routine removal. However, “no symptoms” doesn’t always mean “no problem.”
Should Asymptomatic Implants Be Removed?
This is a deeply personal decision. On one hand, the risk of developing BIA-ALCL is statistically low (less than 0.03% for BIOCELL implants). On the other hand, for a cancer survivor or someone with a BRCA mutation, even a small risk is too much.
We recommend:
- Regular Monitoring: If you keep the implants, you should have an MRI every 2-3 years to check for “silent” ruptures or fluid.
- Risk-Benefit Analysis: Consider the risks of a new surgery versus the peace of mind of being “flat” or having smooth implants.
- Legal Protection: Even if you choose to wait, you should document your implant type now. Joining a breast cancer class action lawsuit investigation can help protect your future rights.
Options for Reconstruction and Revision
If you decide to proceed with removal, you aren’t without options. Many women choose to transition to safer alternatives:
- Smooth Silicone or Saline Implants: These have no known link to BIA-ALCL.
- Fat Grafting: Using your own fat to provide a natural volume increase without a foreign object.
- Explant Only: Choosing to remain without implants, often combined with a “breast lift” for aesthetic results.
When undergoing removal, most experts recommend an “en-bloc” or total capsulectomy to ensure all potentially contaminated tissue is removed. You can find more info about breast implant services and surgical options through our network of experts.
Frequently Asked Questions about Gummy Bear Implant Recalls
Does the recall mean my implants are definitely going to cause cancer?
No. The vast majority of women with these implants will never develop BIA-ALCL. The recall was a preventative measure because the relative risk was significantly higher than other models, not because the absolute risk is high for every individual.
Will Allergan pay for my removal surgery if I don’t have symptoms?
Generally, no. Allergan’s warranty program typically only covers the cost of the replacement device itself. They usually only cover surgical fees if there is a confirmed diagnosis of BIA-ALCL. This is exactly why many women are turning to the legal system to recover those out-of-pocket costs.
How long do I have to file a legal claim for compensation?
This depends on the “Statute of Limitations” in your state. In many cases, the clock starts ticking from the moment you discovered (or should have discovered) the injury or the link to the recall. Because these laws vary wildly across the U.S., it is best to speak with an attorney as soon as possible.
Conclusion
The recall on gummy bear implants has left hundreds of thousands of women in a state of uncertainty. Whether you are currently experiencing symptoms or are simply worried about the long-term health implications of the BIOCELL texture, you deserve clear answers and fair treatment.
At Tort Advisor, we connect clients with top-rated specialty attorneys who have proven results in complex personal injury and product liability cases. We believe that no woman should have to choose between her financial stability and her physical safety.
If you have been affected by the Allergan recall, don’t navigate this alone. Use our breast implant settlement calculator to explore your options and take the first step toward the advocacy and compensation you deserve.
Free Confidential Case Evaluation
Complete the short form below to get an immediate FREE case review with an expert in your specific claim. Don't wait, your case could be time sensitive to file a claim.
Related Posts
Institutional sexual abuse lawsuit 2026: Churches, schools, hospitals, detention centers. Who qualifies, settlement amounts, how to file. Free confidential review. 1 (855) 664-8713.
SSDI claims 2026: Complete guide to winning Social Security Disability benefits. Who qualifies, how to appeal denials, free case review. 1 (855) 664-8713.
Who qualifies for WTC VCF claims 2026? FDNY, NYPD, first responders — 50+ cancers covered. Tax-free awards. Free review. 1 (855) 664-8713.
World Trade Center lawsuit 2026: 9/11 VCF guide for FDNY, NYPD, first responders. 50+ cancers covered. Free VCF review. 1 (855) 664-8713.
Lyft sexual assault lawsuit 2026: 2,000+ reported incidents. Who qualifies, settlement amounts, how to sue Lyft. Free confidential review. 1 (855) 664-8713.
Uber sexual assault lawsuit 2026: $8.5M verdict won. Who qualifies, settlement amounts, how to file. Free confidential case review. 1 (855) 664-8713.